Trusted for safety.
Chosen for outcomes.
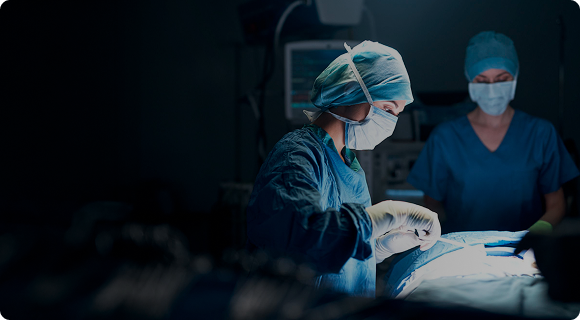
Our biosurgery business is at the forefront of medical innovation with our flagship hemostasis product, the TachoSil® Fibrin Sealant Patch, a vital tool in surgical procedures. TachoSil is part of Corza Medical’s mission to advance surgical care.Trusted in over 13 million cases, TachoSil delivers faster hemostasis, reduces complications, and lowers treatment costs, making it the preferred choice in a wide range of surgical procedures. Because in critical moments, trust matters.


TachoSil–The gold standard in surgical excellence.
The ready-to-use solution for hemostasis. TachoSil combines the bioactive mechanism of action of human fibrinogen and thrombin with simple handling to support surgeons when every second counts. It activates on contact with blood or fluid, delivering fast and effective control of bleeding. No need for mixing or extra preparation–just apply, press, and proceed with confidence.
The vital function of hemostasis.
Hemostasis is the body’s rapid, tightly regulated response to vessel injury. First, injured vessels constrict and platelets stick together to form a temporary plug (primary hemostasis). Next, the coagulation cascade (secondary hemostasis) activates a proteolytic chain that converts fibrinogen into fibrin, which creates a strong mesh around the platelet plug to stabilize the clot and allow tissue repair. Fibrin is essential, as it transforms a fragile platelet patch into a durable scaffold that resists blood flow and supports wound healing.
The impact of bleeding on hospital costs.
69%
81%
83%
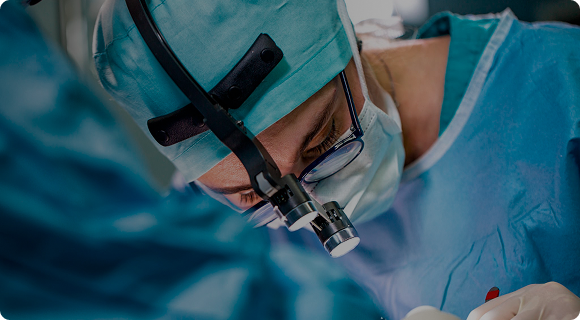
Benefits of TachoSil in surgical procedures.1
28%
14%
As surgical practice increasingly incorporates minimally invasive and robotic assisted techniques, maintaining effective hemostasis remains essential. TachoSil may be used as an adjunct in these procedures when standard surgical techniques are insufficient. Its use can support intraoperative management during minimally invasive and robotic surgery, including in complex cases, with the aim of contributing to controlled surgical outcomes and postoperative recovery.



Ready. Steady. Go.
Easy to use, with no need for mixing or preparation. TachoSil helps reduce complications, lowers intraoperative risks, and supports faster decision-making—so you can operate with more confidence and focus on what truly matters: your patient.
One innovation.
Versatile surgical applications.
Discover how TachoSil brings multiple solutions across surgical specialties, helping surgeons achieve reliable hemostasis with confidence.
Ready for
the future.
Success in our blood. Innovation in our mind.
Since its launch, TachoSil has undergone continuous development expanding its clinical applications, refining usability, and proving its value through extensive studies. From cardiovascular to liver and pediatric surgery, the fibrin sealant patch has become a trusted tool in the hands of surgeons worldwide. Its evolution reflects an ongoing commitment to innovation, patient safety, and surgical efficiency.
References
2: Summary of Product Characteristics TachoSil.
*Last Periodic Benefit Risk Evaluation Report (PBRER) for the United States (US) submission (FDA) Period: 09-Jun-2024 to 08-Jun-2025
COR-BS26-0001-R0
Limitations for Use: TachoSil cannot safely or effectively be used in place of sutures or other form of mechanical ligation for the treatment of major arterial or venous bleeding. • Not for use in children under one month of age.
Dosage & Administration: For topical use on cardiovascular or hepatic tissue only • Determine the number of patches to be applied by the size of the bleeding area. • Apply the yellow, active side of the patch to the bleeding area. • When applying TachoSil, do not exceed the maximum number of patches. Refer to the Full Prescribing Information for details on administration and maximum number of patches.
Contraindications: Do not apply TachoSil intravascularly. Intravascular application of TachoSil may result in life threatening thromboembolic events. • Do not use TachoSil in individuals known to have anaphylactic or severe systemic reaction to human blood products or horse proteins.
Warnings & Precautions:
Thrombosis. Thrombosis can occur if TachoSil is applied intravascularly. Ensure that TachoSil is applied to the surface of cardiac, vascular, or hepatic tissue only.
Hypersensitivity Reactions. Hypersensitivity or allergic/anaphylactoid reactions may occur in patients receiving TachoSil for the first time or may increase with repetitive applications.
Infection. Avoid application to contaminated or infected areas of the body, or in the presence of active infection.
Adhesions. TachoSil contains collagen, which may adhere to bleeding surfaces. May carry a risk of gastrointestinal obstruction in abdominal surgery due to tissue adhesions. To prevent the development of tissue adhesions at undesired sites, ensure tissue areas outside the desired application area are adequately cleansed before administration of TachoSil.
Compression. Avoid packing in cavities or closed spaces because this may cause compression of underlying tissue.
Non-Adherence or Dislodged Material. Use only the minimum number of TachoSil patches necessary to achieve hemostasis. Do not pack. Remove unattached pieces of TachoSil.
Transmissible Infectious Agents. May carry a risk of transmitting infectious agents, such as viruses, and theoretically, the variant Creutzfeldt-Jakob disease (vCJD) agent and the Creutzfeldt-Jakob disease (CJD) agents.
Use in specific populations: Pediatric Use: Use of TachoSil in children under the age of one month may be unsafe or ineffective due to small size and limited ability to apply the patch as recommended.
Adverse Reactions: The adverse reactions reported in more than 1% of patients during clinical trials were anemia, nausea and vomiting, fever, abdominal pain, increased white blood cell count, ascites, itching, atrial fibrillation, pleural effusion, gastrointestinal hemorrhage, wound infection, hypophosphatemia, urinary tract infection, and post-procedural bile leakage in hepatic surgery. Refer to the Full prescribing Information for details on, contraindications, warnings, precautions, and adverse reactions.
Marketing Authorisation Holder: Corza Medical GmbH, Speditionstrasse 21, 40221 Düsseldorf, Germany. US License no. 2294.Revision date: 08/2025
A trusted name worldwide.
Used in operating rooms around the world, TachoSil stands for reliability, safety, and consistent surgical performance—wherever it’s needed.
