TachoSil®–The gold standard in surgical excellence.
The ready-to-use
solution for hemostasis
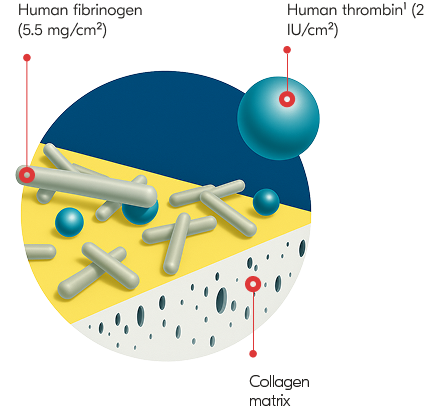
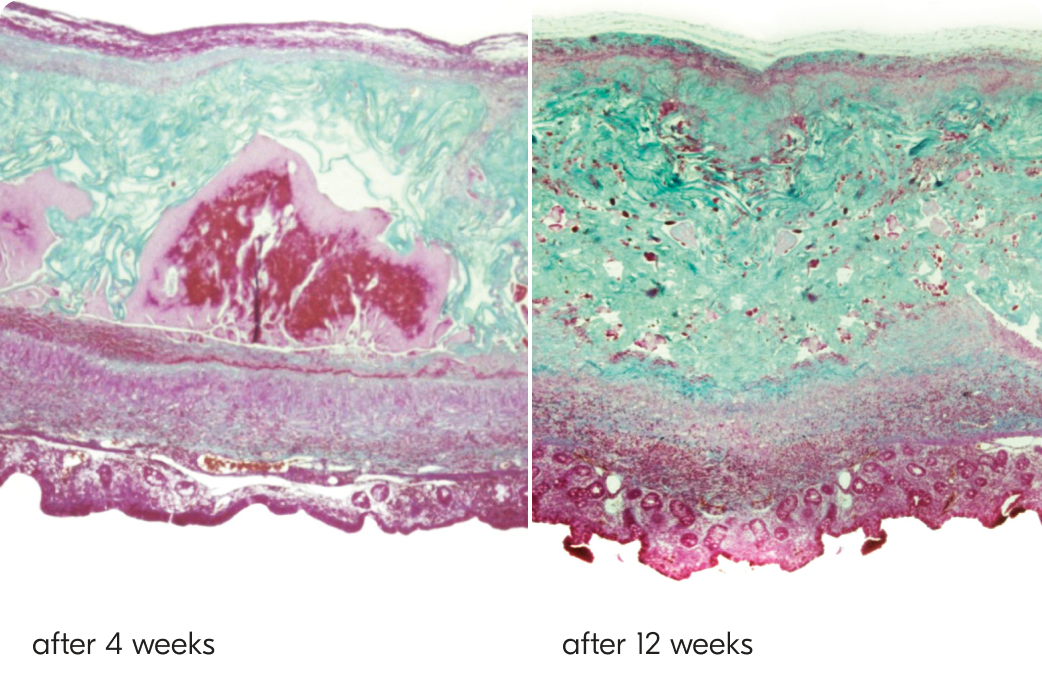
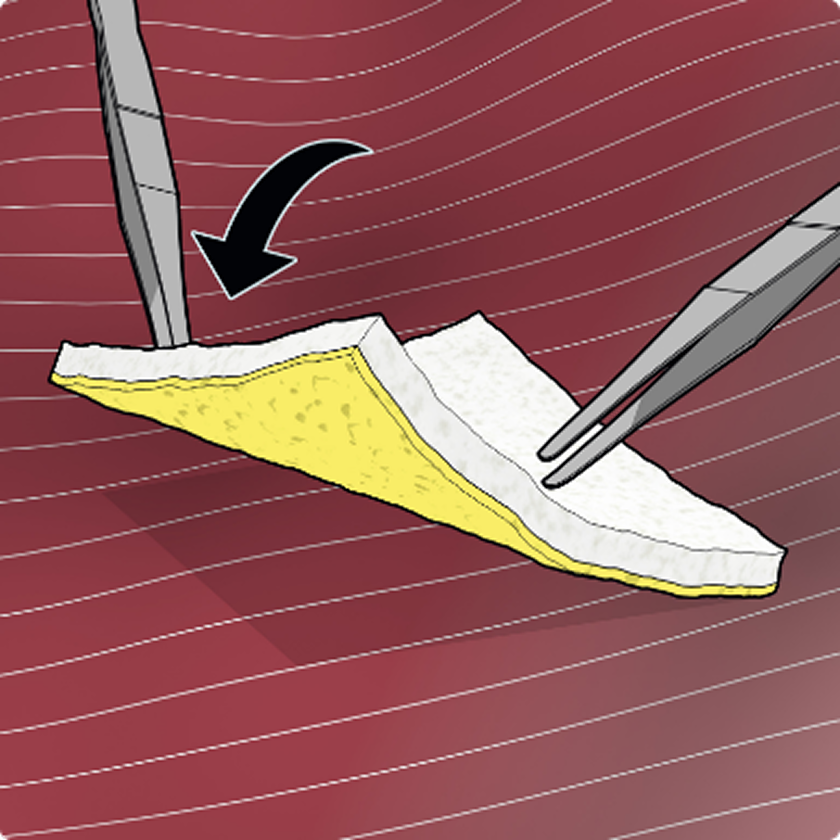
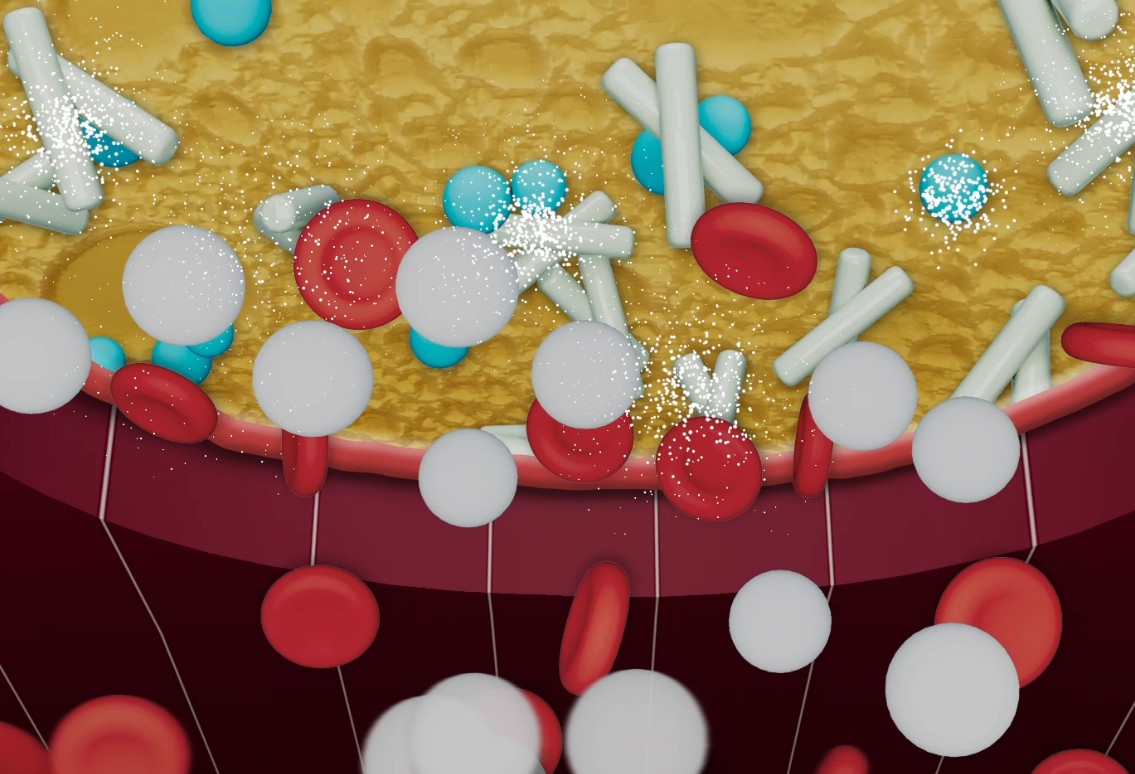
TachoSil is a fibrin sealant patch coated with fibrinogen and thrombin that activates on contact with blood to deliver fast, effective control of bleeding. With proven efficacy across open and minimally invasive procedures, it combines mechanical strength with physiologic hemostasis. Developed from TachoComb and trusted since 2004.
Own the moment
of responsibility.
Minimally invasive. Maximum control. Reliable hemostasis in robotic surgery.
TachoSil seamlessly integrates into minimally invasive and open workflows, combining trusted performance with precision.

TachoSil comes in a double sterile package. The folding box contains the package leaflet and an aluminium composite foil container. Air and thus moisture are kept out by this heat-sealed aluminium foil. This ensures that fibrinogen and thrombin do not react with each other prematurely and the full adhesive strength remains until it is applied.
Downloads
View TachoSil in action.


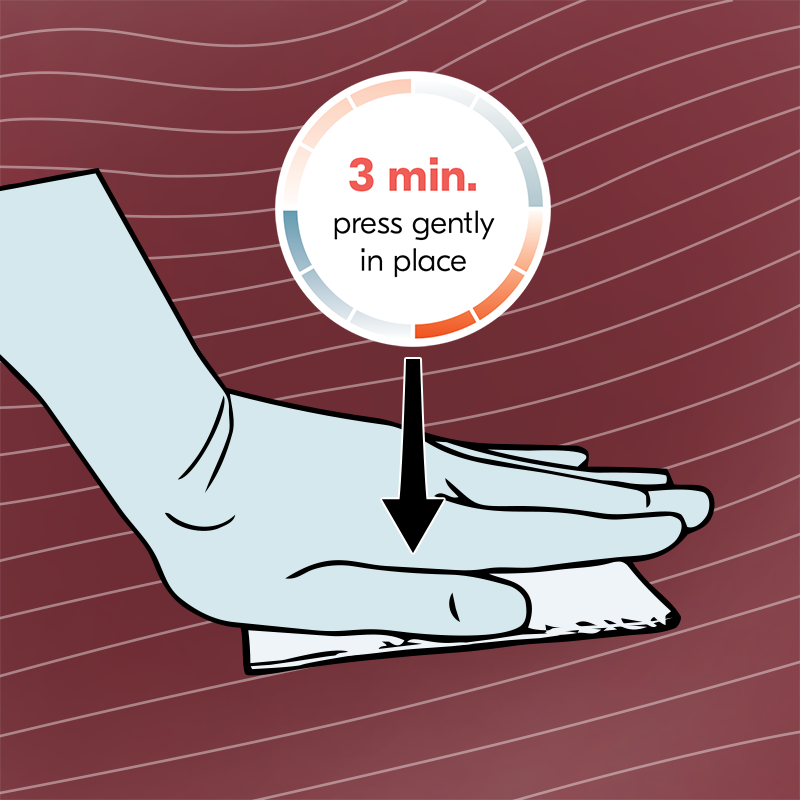
Ready. Steady. Go.
Easy to use, with no need for mixing or preparation. TachoSil helps reduce complications, lowers intraoperative risks, and supports faster decision-making—so you can operate with more confidence and focus on what truly matters: your patient.
Ready for
the future.
Precision in every layer.
Safety in every step.
Innovation made in Austria since 1992: TachoSil has been manufactured in Linz since 2004, building on the legacy of TachoComb. Produced in class C cleanrooms, it meets the highest pharmaceutical quality standards.
Note: The information in this manufacturing video is not country specific and may vary from the approved product information in the country where you are located. Not all sizes and formulations of TachoSil are available in all markets.

A trusted name worldwide.
Used in operating rooms around the world, TachoSil stands for reliability, safety, and consistent surgical performance—wherever it’s needed.

References
COR-BS26-0001-R0
Limitations for Use: TachoSil cannot safely or effectively be used in place of sutures or other form of mechanical ligation for the treatment of major arterial or venous bleeding. • Not for use in children under one month of age.
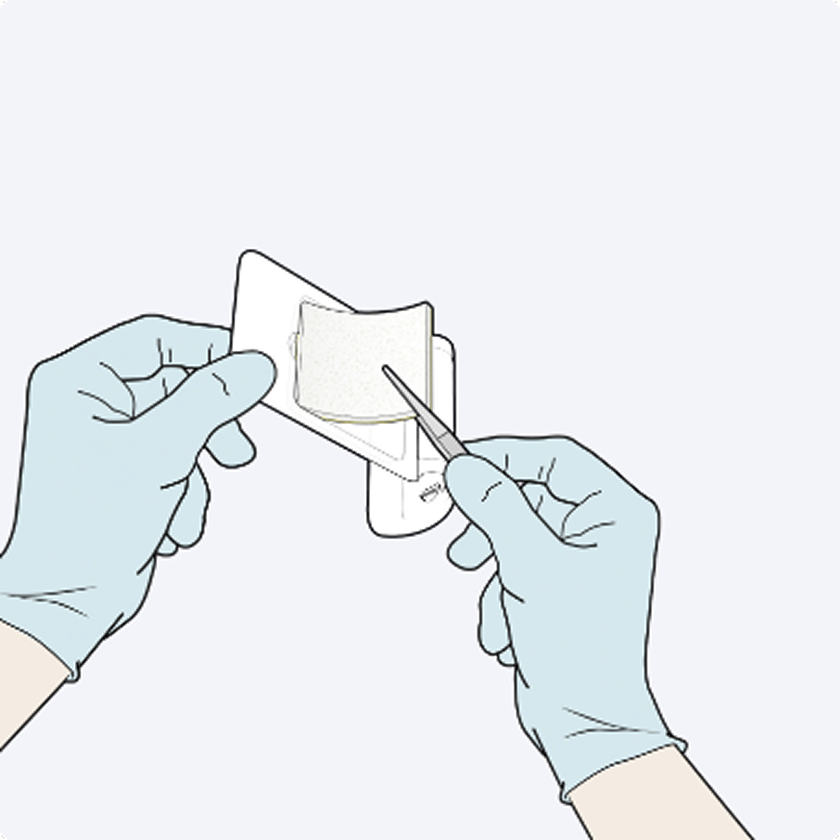
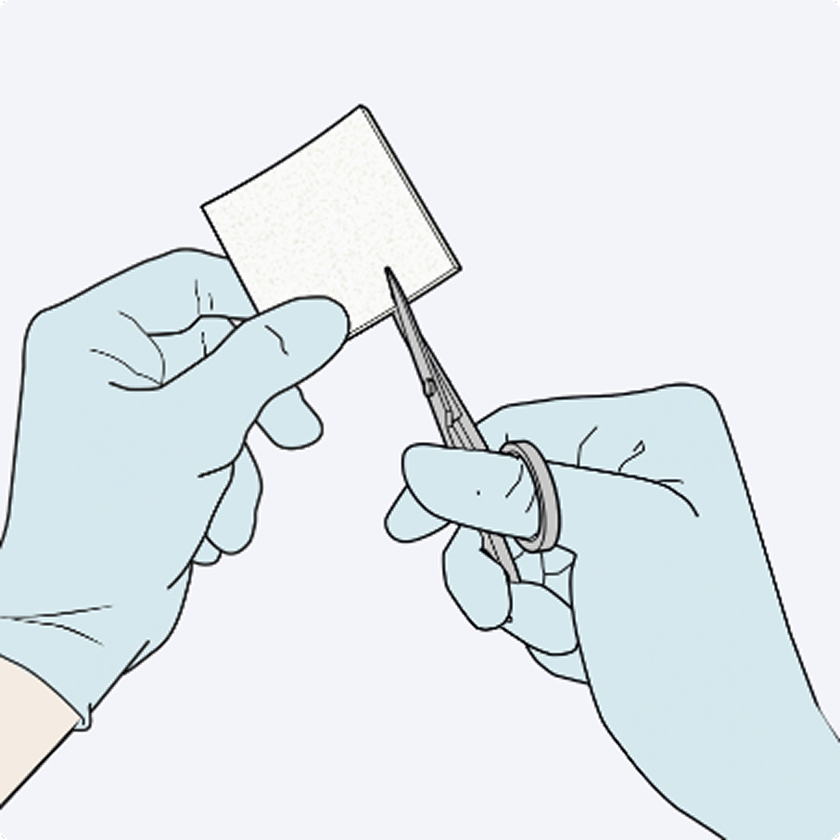
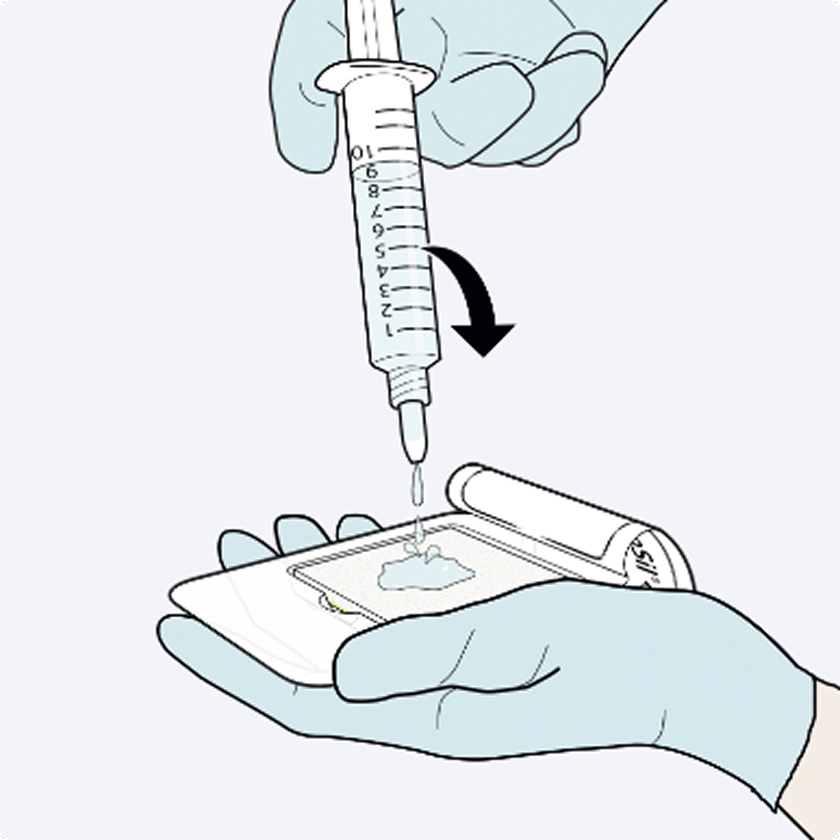
Dosage & Administration: For topical use on cardiovascular or hepatic tissue only • Determine the number of patches to be applied by the size of the bleeding area. • Apply the yellow, active side of the patch to the bleeding area. • When applying TachoSil, do not exceed the maximum number of patches. Refer to the Full Prescribing Information for details on administration and maximum number of patches.
Contraindications: Do not apply TachoSil intravascularly. Intravascular application of TachoSil may result in life threatening thromboembolic events. • Do not use TachoSil in individuals known to have anaphylactic or severe systemic reaction to human blood products or horse proteins.
Warnings & Precautions:
Thrombosis. Thrombosis can occur if TachoSil is applied intravascularly. Ensure that TachoSil is applied to the surface of cardiac, vascular, or hepatic tissue only.
Hypersensitivity Reactions. Hypersensitivity or allergic/anaphylactoid reactions may occur in patients receiving TachoSil for the first time or may increase with repetitive applications.
Infection. Avoid application to contaminated or infected areas of the body, or in the presence of active infection.
Adhesions. TachoSil contains collagen, which may adhere to bleeding surfaces. May carry a risk of gastrointestinal obstruction in abdominal surgery due to tissue adhesions. To prevent the development of tissue adhesions at undesired sites, ensure tissue areas outside the desired application area are adequately cleansed before administration of TachoSil.
Compression. Avoid packing in cavities or closed spaces because this may cause compression of underlying tissue.
Non-Adherence or Dislodged Material. Use only the minimum number of TachoSil patches necessary to achieve hemostasis. Do not pack. Remove unattached pieces of TachoSil.
Transmissible Infectious Agents. May carry a risk of transmitting infectious agents, such as viruses, and theoretically, the variant Creutzfeldt-Jakob disease (vCJD) agent and the Creutzfeldt-Jakob disease (CJD) agents.
Use in specific populations: Pediatric Use: Use of TachoSil in children under the age of one month may be unsafe or ineffective due to small size and limited ability to apply the patch as recommended.
Adverse Reactions: The adverse reactions reported in more than 1% of patients during clinical trials were anemia, nausea and vomiting, fever, abdominal pain, increased white blood cell count, ascites, itching, atrial fibrillation, pleural effusion, gastrointestinal hemorrhage, wound infection, hypophosphatemia, urinary tract infection, and post-procedural bile leakage in hepatic surgery. Refer to the Full prescribing Information for details on, contraindications, warnings, precautions, and adverse reactions.
Marketing Authorisation Holder: Corza Medical GmbH, Speditionstrasse 21, 40221 Düsseldorf, Germany. US License no. 2294.Revision date: 08/2025