TachoSil® in neurosurgical procedures.
Discover the far-reaching potential of TachoSil in the specialty of neurosurgery.
Reliable hemostasis
and sealing in
neurosurgery.
TachoSil provides both hemostatic and sealing properties, creating a liquid-tight barrier over the dural suture line.
TachoSil is indicated in adults for supportive sealing of the dura mater to prevent postoperative cerebrospinal leakage following neurological surgery.1
Own the moment
of responsibility.
Proven benefits of TachoSil in Neurosurgery
Versatile application in skull base, transsphenoidal, minimally invasive spine, and complex cranial procedures.
- Observational studies and systematic reviews highlight its use in high-risk neurosurgical settings2,7,8.
Reduces CSF leak risk after cranial and spinal dural closure, including skull base and spine surgery.
- Multicentre RCT in 726 skull base patients showed low leak rates (6.9% vs 8.2%) and confirmed safety8.
- Retrospective spine cohort (n=85) reported 89.7% success with TachoSil monotherapy 9.
Effective in incidental durotomy repair, reducing need for reoperation.
- Observational study (n = 55) found no reoperations for dural closure failure and only 1.8% readmission due to CSF leak3
- Retrospective spine surgery patient cohort (n = 85) reported 89.7% success with TachoSil monotherapy9
- A one-year follow-up cohort of small dural tears repaired with a fibrin-sealant-coated collagen patch reported no postoperative CSF leaks at one year10
Safe and well-tolerated, with no product-related adverse events reported.
- RCT and long-term follow-up studies confirm absence of granuloma or infection risk2,7,8.
View TachoSil in action.
TachoSil application to restore dura mater loss resulting from the removal of a spheno-orbital meningioma by Prof. dr hab. n. med. Piotr Ładziński
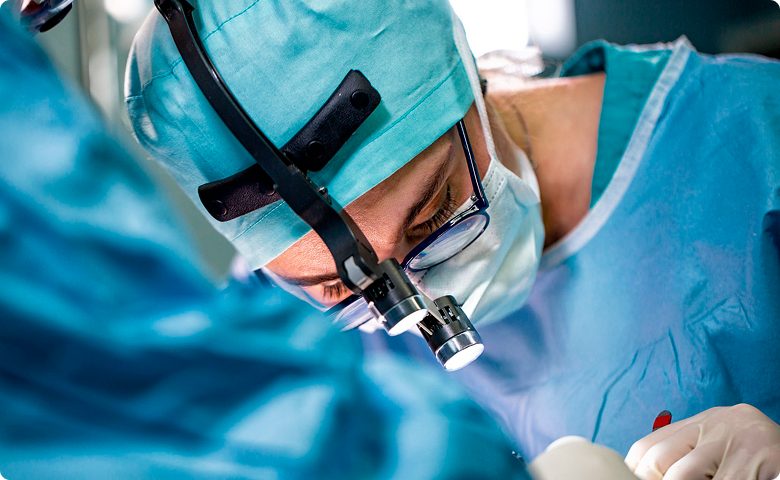

More confidence. Better outcomes.
References
1. Summary of Product Characteristics TachoSil. EMA. June 2025.
2. Baussart B, Venier A, Jouinot A, Reuter G, Gaillard S. Closure strategy for endoscopic pituitary surgery: Experience from 3015 patients. Front Oncol. 2023 Jan 4;12:1067312. doi: 10.3389/fonc.2022.1067312. PMID: 36686774; PMCID: PMC9846073.
3. Chaliparambil RK, Kemeny HR, Mukherjee S, Krushelnytskyy M, Wolinksy JP, Swong K, et al. Use of TachoSil for durotomy repair in spine surgery: a single-center retrospective review. Neurosurg Focus. 2025 Feb 1;58(2):E12.
4. Papavero L, Engler N, Kothe R. Incidental durotomy in spine surgery: first aid in ten steps. Eur Spine J. 2015 Sep;24(9):2077-84. doi: 10.1007/s00586-015-3837-x. Epub 2015 Mar 4. PMID: 25735610.
5. Trathitephun W, Asawasaksakul A, Jaruwanneechai K, Pakdeenit B, Suebsing A, Liu Y, Kim JS, Suvithayasiri S. Intraoperative Management of Iatrogenic Durotomy in Endoscopic Spine Surgery: A Systematic Review. Neurospine. 2024 Sep;21(3):756-766. doi: 10.14245/ns.2448346.173. Epub 2024 Sep 30. PMID: 39363456; PMCID: PMC11456941.
6. Papavero L, Engler N, Kothe R. Incidental durotomy in spine surgery: first aid in ten steps. Eur Spine J. 2015 Sep;24(9):2077-84. doi: 10.1007/s00586-015-3837-x. Epub 2015 Mar 4. PMID: 25735610.
7. Kamenova M, Leu S, Mariani L, Schaeren S, Soleman J. Management of Incidental Dural Tear During Lumbar Spine Surgery. To Suture or Not to Suture? World Neurosurg. 2016 Mar;87:455–62.
8. George B, Matula C, Kihlström L, Ferrer E, Tetens V. Safety and Efficacy of TachoSil (Absorbable Fibrin Sealant Patch) Compared With Current Practice for the Prevention of Cerebrospinal Fluid Leaks in Patients Undergoing Skull Base Surgery: A Randomized Controlled Trial. Neurosurgery. 2017 Jun 1;80(6):847–53.
9. Delgado-López, P.D., Barreras-García, A., Herrero Gutiérrez, A.S. et al. Efficacy of a synthetic collagen-based sealant (TachoSil) in preventing cerebrospinal fluid leak following planned and incidental durotomies in spine surgery: a retrospective cohort study. Neurosurg Rev 49, 4 (2026). https://doi.org/10.1007/s10143-025-03917-z
10. Galarza M, Gazzeri R, Alfaro R, de la Rosa P, Arraez C, Piqueras C. Evaluation and management of small dural tears in primary lumbar spinal decompression and discectomy surgery. J Clin Neurosci. 2018 Apr;50:177–82.
2. Baussart B, Venier A, Jouinot A, Reuter G, Gaillard S. Closure strategy for endoscopic pituitary surgery: Experience from 3015 patients. Front Oncol. 2023 Jan 4;12:1067312. doi: 10.3389/fonc.2022.1067312. PMID: 36686774; PMCID: PMC9846073.
3. Chaliparambil RK, Kemeny HR, Mukherjee S, Krushelnytskyy M, Wolinksy JP, Swong K, et al. Use of TachoSil for durotomy repair in spine surgery: a single-center retrospective review. Neurosurg Focus. 2025 Feb 1;58(2):E12.
4. Papavero L, Engler N, Kothe R. Incidental durotomy in spine surgery: first aid in ten steps. Eur Spine J. 2015 Sep;24(9):2077-84. doi: 10.1007/s00586-015-3837-x. Epub 2015 Mar 4. PMID: 25735610.
5. Trathitephun W, Asawasaksakul A, Jaruwanneechai K, Pakdeenit B, Suebsing A, Liu Y, Kim JS, Suvithayasiri S. Intraoperative Management of Iatrogenic Durotomy in Endoscopic Spine Surgery: A Systematic Review. Neurospine. 2024 Sep;21(3):756-766. doi: 10.14245/ns.2448346.173. Epub 2024 Sep 30. PMID: 39363456; PMCID: PMC11456941.
6. Papavero L, Engler N, Kothe R. Incidental durotomy in spine surgery: first aid in ten steps. Eur Spine J. 2015 Sep;24(9):2077-84. doi: 10.1007/s00586-015-3837-x. Epub 2015 Mar 4. PMID: 25735610.
7. Kamenova M, Leu S, Mariani L, Schaeren S, Soleman J. Management of Incidental Dural Tear During Lumbar Spine Surgery. To Suture or Not to Suture? World Neurosurg. 2016 Mar;87:455–62.
8. George B, Matula C, Kihlström L, Ferrer E, Tetens V. Safety and Efficacy of TachoSil (Absorbable Fibrin Sealant Patch) Compared With Current Practice for the Prevention of Cerebrospinal Fluid Leaks in Patients Undergoing Skull Base Surgery: A Randomized Controlled Trial. Neurosurgery. 2017 Jun 1;80(6):847–53.
9. Delgado-López, P.D., Barreras-García, A., Herrero Gutiérrez, A.S. et al. Efficacy of a synthetic collagen-based sealant (TachoSil) in preventing cerebrospinal fluid leak following planned and incidental durotomies in spine surgery: a retrospective cohort study. Neurosurg Rev 49, 4 (2026). https://doi.org/10.1007/s10143-025-03917-z
10. Galarza M, Gazzeri R, Alfaro R, de la Rosa P, Arraez C, Piqueras C. Evaluation and management of small dural tears in primary lumbar spinal decompression and discectomy surgery. J Clin Neurosci. 2018 Apr;50:177–82.
TACHOSIL SmPC
Links to regional SMPC:
Abbreviated SmPC:
TachoSil Sealant Matrix (5.5 mg per cm2 of human fibrinogen, 2.0 IU per cm2 of human thrombin)
Statement: Before prescribing, consult/refer to the full prescribing information. Presentation: An off-white sealant matrix. The active side of the matrix is coated with fibrinogen and thrombin, is marked by a yellow colour. Supplied, ready to use, in sterile packaging. Legal Classification: Restricted prescription only medicine. Indications: In adults and children from 1 month old, for supportive treatment in surgery for improvement of haemostasis, to promote tissue sealing, and for suture support in vascular surgery where standard techniques are insufficient; also, in adults for supportive sealing of the dura mater to prevent postoperative cerebrospinal leakage following neurosurgical procedures. Dosage & Administration: For epilesional use only. Use is restricted to experienced surgeons. The quantity to be applied is governed by the size of wound area, and the underlying clinical need for the patient. In clinical studies, the individual dosages have typically ranged from 1-3 units (9.5 cm x 4.8 cm); application of up to 10 units has been reported. For smaller wounds, the smaller size matrices (4.8 cm x 4.8 cm or 3.0 cm x 2.5 cm) or the pre-rolled matrix (based on a matrix of 4.8 cm x 4.8 cm) is recommended. TachoSil should be used under sterile conditions and immediately after opening the inner sterile cover. Prior to application, the wound area should be cleansed, e.g. from blood, disinfectants and other fluids. For Flat TachoSil, the sterile package should be pre-moistened in saline solution and applied immediately. The yellow, active side of the matrix is applied to the bleeding/leaking surface and held against it with a gentle pressure for 3-5 minutes. For pre-rolled TachoSil, after removing from the sterile package, it should be applied immediately through the trocar without pre-moistening. The yellow, active side of the matrix is applied to the bleeding/leaking surface using e.g., a pair of cleansed forceps and held against it with a moist pad under gentle pressure for 3-5 minutes. Pressure is applied with moistened gloves or a moist pad. Avoid TachoSil sticking to surgical instruments, gloves or adjacent tissues covered with blood by cleansing them before application. After pressing TachoSil to the wound, the glove or the pad must be removed carefully. To avoid TachoSil from being pulled loose it may be held in place at one end, e.g. with a pair of forceps. In the case of stronger bleeding, it may be applied without pre-moistening, while also pressing gently to the wound for 3-5 minutes. The active side of TachoSil should be applied so that it extends 1-2 cm beyond the margins of the wound. If more than one matrix is used, they should overlap. TachoSil can be cut to the correct size and shaped if too large. In neurosurgery, TachoSil should be applied on top of the primary dura closure. Contraindications: Intravascular use; hypersensitivity to the active substances or to any of the excipients. Warnings & Precautions: No specific data available on the use of this product in gastrointestinal anastomoses surgery. Life threatening thromboembolic complications may occur if the preparation is applied intravascularly. Allergic type hypersensitivity reactions are possible, as with any protein product. If hypersensitivity reactions occur, the administration must be discontinued immediately. To prevent the development of tissue adhesions at undesired sites, ensure tissue areas outside the desired application area are adequately cleansed before administration. In the case of shock, the current medical standards for shock treatment should be followed. Standard measures to prevent infections resulting from the use of medicinal products prepared from human blood or plasma include selection of donors, screening of individual donations and plasma pools for specific markers of infection and the inclusion of effective manufacturing steps for the inactivation/removal of viruses. Measures taken are considered effective for enveloped viruses such as HIV, HBV and HCV and for the non-enveloped virus HAV. Measures may be of limited value against non-enveloped viruses such as parvovirus B19. Parvovirus B19 infection may be serious for pregnant women (foetal infection) and for individuals with immunodeficiency or increased erythropoiesis (e.g., haemolytic anaemia). It is recommended to record the name and the batch number of the product administered to the patient. Some cases of product non-adhesion issues have been reported in the form of lack of product adhesion / lack of efficacy. Correct product handling and application is required Interactions: No interaction studies have been performed. Similar to comparable products or thrombin solutions, the sealant may be denatured after exposure to solutions containing alcohol, iodine, or heavy metals. Such substances should be removed to the greatest possible extent before applying the sealant. Fertility, Pregnancy & Lactation: Safety for use in human pregnancy or breastfeeding has not been established in the clinical studies. Only administer to pregnant and breastfeeding women if clearly needed. Effects on Ability to Drive and Use Machines: Not relevant. Undesirable Effects: Hypersensitivity or allergic reactions (in isolated cases these reactions may progress to severe anaphylaxis; some cases of product residue causing granuloma), thromboembolic complications may occur if used intravascularly, and adhesions and intestinal obstruction when used in abdominal surgery. Refer to the SmPC for details on full side effect profile and interactions. Overdose information: No case of overdose has been reported. Interactions with Other Medicinal Products: No interaction studies have been performed. Similar to comparable products or thrombin solutions, the sealant may be denatured after exposure to solutions containing alcohol, iodine or heavy metals (e.g., antiseptic solutions). Such substances should be removed to the greatest possible extent before applying the sealant. Use in Special Populations: Limited data are available to support efficacy and safety of TachoSil in the paediatric population. In clinical studies, a total of 36 paediatric patients aged 0-13 years were treated with TachoSil in hepatic surgery. Pack Sizes: Package with 1 matrix of 9.5 cm x 4.8 cm
Package with 2 matrices of 4.8 cm x 4.8 cm
Package with 1 matrix of 3.0 cm x 2.5 cm
Package with 5 matrices of 3.0 cm x 2.5 cm
Package with 1 pre-rolled matrix of 4.8 cm x 4.8 cm
Not all pack sizes may be marketed.
Marketing Authorisation Holder: Corza Medical GmbH, Speditionstraße 21, 40221 Düsseldorf, Germany.
The full SmPC can be obtained from Corza Medical GmbH.
Marketing Authorisation Numbers: EU/1/04/277/001-005.
Date of Revision of the Text: 8 May 2025.
Link/QR to Full SmPC or Prescribing information.
Need help finding something?
Our team is here to help.


