New study shows no clear benefit for Triclosan coated sutures
WHO 2018 24 and CDC 25 guidelines conditionally recommend the use of Triclosan(TCS)-coated sutures to reduce SSI rates, yet these recommendations are based on low-to-moderate quality results according to GRADE* with many having conflicts of interest. 26
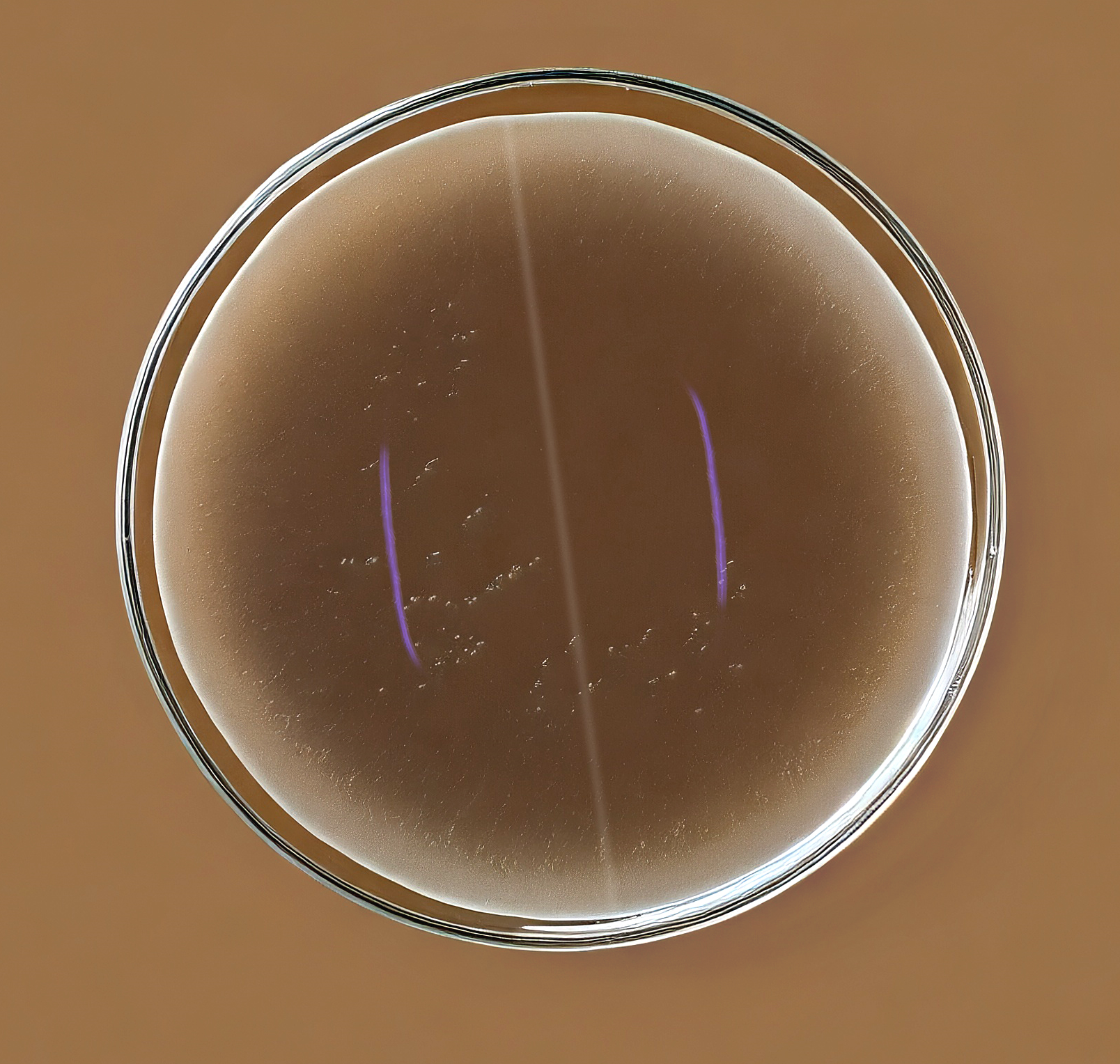
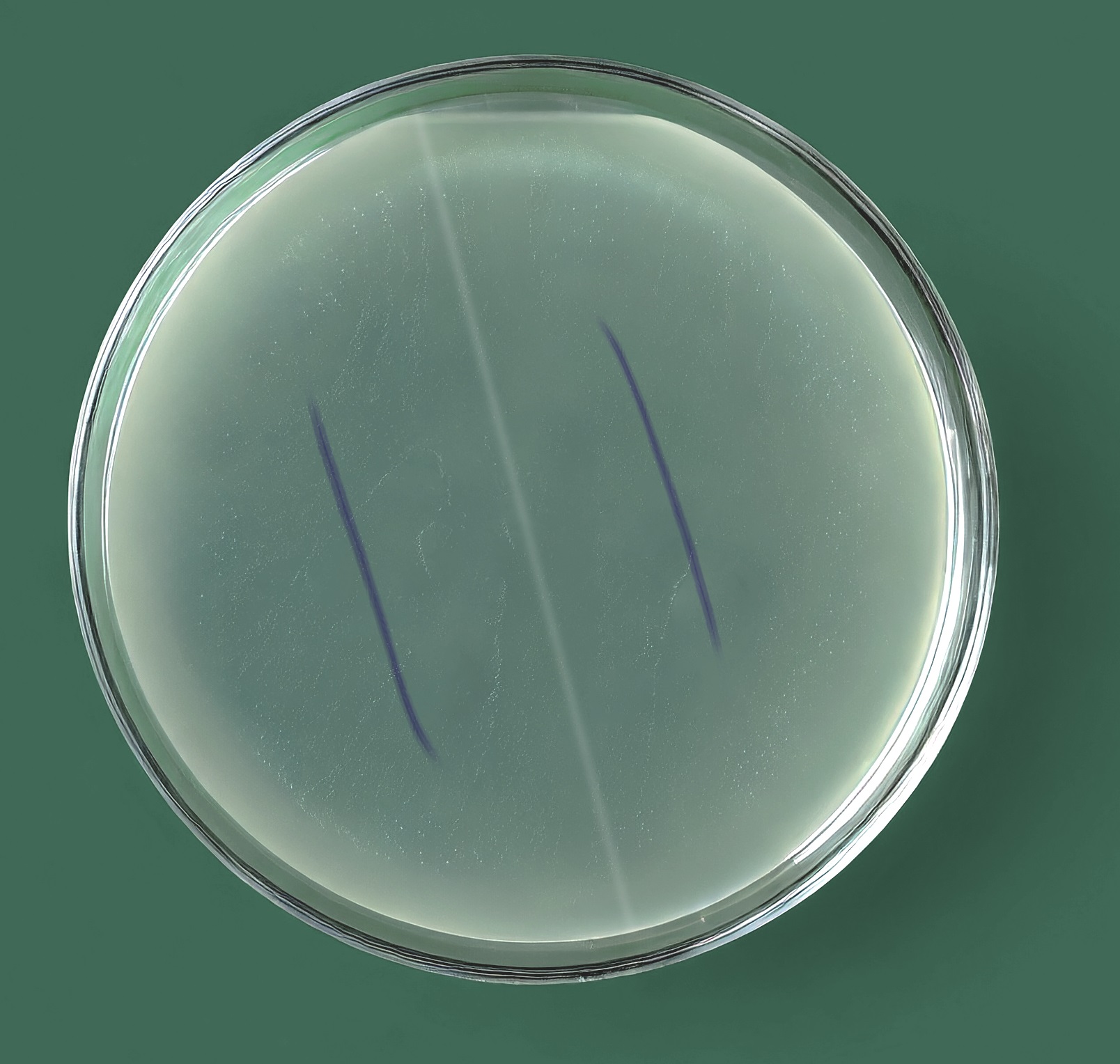
Image 1: No zone of inhibition around coated and uncoated suture against Enterococcus spp; and Pseudomonas spp; adaptation of image, property of SenGupta et. all 2014
The in vitro experiments revealed that triclosan coated sutures, hailed for their antimicrobial properties, exhibited no inhibitory effects against Pseudomonas spp. and Enterococcus spp. isolates, crucial causative agents of post-operative wound infections.1,2
Moreover, TCS could interact antagonistically with other antibiotics against certain bacteria, potentially fostering cross-resistance.2
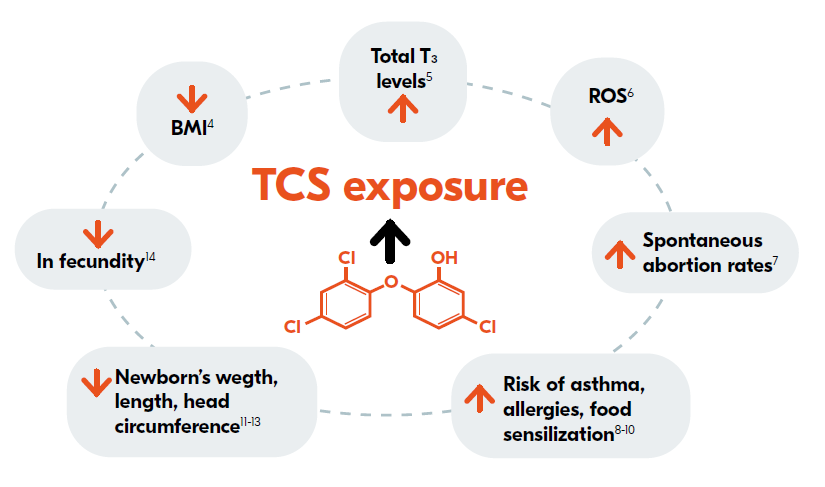
Image 2: Human epidemiology studies show an association of an increase in urinary triclosan concentrations with a variety of detrimental endpoints.15 Based on the figure of the publication of Lisa M. Weatherly et al. 2017. ROS: Reactive Oxygen Species
- It has been detected in human milk samples and in urine at high concentrations.16
- Interferes with body’s thyroid hormone metabolism and may be a potential endocrine disruptor.16
- There are concerns about its link with highly carcinogenic dioxin.16
- Many studies observed an association between a rise in TCS exposure and reproductive and developmental defects in infants.15
- FDA, EE, Japan and some Asian countries banned its use in households and consumer products.17-22
- TCS is associated with a wide array of adverse ecotoxicological effects across various species and has been detected in bodies of water globally, with studies revealing its prevalence in streams, rivers, and lakes, indicating its pervasive environmental presence and potential ecological impact.15
- The widespread presence of TCS in various water sources elevates human non-voluntary exposure to the compound.3
FALCON study, conducted from the National Institute of Health Research Unit on Global Surgery in 2021,
KEY POINTS
Why was FALCON initiated?
In 2016, WHO27 made 29 recommendations for preventing SSI, mostly based on low to moderate quality studies. They conditionally recommended triclosan-coated sutures based on moderate-quality evidence. Further evaluation was advised for both interventions in lower-resource settings. 26
What are the implications of this new evidence?
Implementing guidelines for SSI prevention in real-world conditions, particularly in resource-limited settings, can be expensive. The FALCON findings suggest that routine use of chlorhexidine and triclosan-coated sutures may not be supported. 26
Are there study limitations?
The FALCON study obtained high-quality data with a low risk of bias, although the absence of barbed sutures represents a limitation of the study. 26
Strengths of FALCON:
This is the largest RCT aimed at reducing SSI in LMICs. It is a multi-country, pragmatic, and low-risk of bias trial. It encompasses a diverse array of procedures and incorporates hard-to-reach patients, ensuring geographical generalizability. 26
FALCON study conclusion
Recently published high-quality data underscore the necessity for a critical review of SSI guidelines, prompting the implementation of more stringent measures globally to mitigate environmental and health risks.
*Grading of Recommendations Assessment, Development and Evaluation
- SenGupta M, et al. In vitro efficacy of triclosan coated polyglactin 910 suture against common bacterial pathogen causing surgical site infection. International Journal of Infection Control [Internet]. 2014 Mar 26 [cited 2024 Jan 10];10(2). Available from: https://ijic.info/article/view/11840.
- Shrestha P, et al. Synergistic and antagonistic interactions of triclosan with various antibiotics in bacteria. Journal of Environmental Science and Health, Part C. 2020 Jul 2;38(3):187–203.
- Milanović M, et al. Comprehensive insight into triclosan-from widespread occurrence to health outcomes. Environ Sci Pollut Res Int. 2023 Feb;30(10):25119–40.
- Li S, et al. Urinary triclosan concentrations are inversely associated with body mass index and waist circumference in the US general population: Experience in NHANES 2003-2010. Int J Hyg Environ Health. 2015 Jun;218(4):401–6.
- Koeppe ES, et al. Relationship between urinary triclosan and paraben concentrations and serum thyroid measures in NHANES 2007-2008. Sci Total Environ. 2013 Feb 15;445–446:299–305.
- Lv Y, et al. Exposure of children to BPA through dust and the association of urinary BPA and triclosan with oxidative stress in Guangzhou, China. Environ Sci Process Impacts. 2016 Dec 8;18(12):1492–9.
- Wang X, et al. Triclosan causes spontaneous abortion accompanied by decline of estrogen sulfotransferase activity in humans and mice. Sci Rep. 2015 Dec 15;5:18252.
- Clayton EMR, et al. The impact of bisphenol A and triclosan on immune parameters in the U.S. population, NHANES 2003-2006. Environ Health Perspect. 2011 Mar;119(3):390–6.
- Spanier AJ, et al. The associations of triclosan and paraben exposure with allergen sensitization and wheeze in children. Allergy Asthma Proc. 2014;35(6):475–81.
- Hong S, et al. Association between exposure to antimicrobial household products and allergic symptoms. Environ Health Toxicol. 2014;29:e2014017.
- Lassen TH, et al. Prenatal Triclosan Exposure and Anthropometric Measures Including Anogenital Distance in Danish Infants. Environ Health Perspect. 2016 Aug;124(8):1261–8.
- Philippat C, et al. Prenatal exposure to phenols and growth in boys. Epidemiology. 2014 Sep;25(5):625–35.
- Etzel TM, et al. Urinary triclosan concentrations during pregnancy and birth outcomes. Environ Res. 2017 Jul;156:505–11.
- Vélez MP, et al. Female exposure to phenols and phthalates and time to pregnancy: the Maternal-Infant Research on Environmental Chemicals (MIREC) Study. Fertil Steril. 2015 Apr;103(4):1011-1020.e2.
- Weatherly LM, Gosse JA. Triclosan exposure, transformation, and human health effects. J Toxicol Environ Health B Crit Rev. 2017;20(8):447–69.
- Beyond Pesticides [Internet]. [cited 2024 Mar 1]. Triclosan: Health Effects. Available from: https://www.beyondpesticides.org/resources/antibacterials/triclosan/health-effects
- Asean bans five parabens, restricts triclosan in cosmetics [Internet]. [cited 2024 Mar 1]. Available from: https://product.enhesa.com/22542/asean-bans-five-parabens-restrictstriclosan-in-cosmetics
- EU authorities back triclosan ban in various products [Internet]. [cited 2024 Mar 1]. Available from: https://product.enhesa.com/18783/eu-authorities-back-triclosan-ban-in-various-products
- Commissioner O of the. FDA. FDA; 2020 [cited 2024 Mar 1]. FDA issues final rule on safety and effectiveness of antibacterial soaps. Available from: https://www.fda.gov/news-events/press-announcements/fda-issues-final-rule-safety-and-effectiveness-antibacterial-soaps
- GODDYN S. Parliamentary question | Triclosan ban | P-006697/2016 | European Parliament [Internet]. [cited 2024 Mar 1]. Available from: https://www.europarl.europa.eu/doceo/document/P-8-2016-006697_EN.html
- cosmeticsdesign.com. 2016 [cited 2024 Mar 1]. Japan bans 19 ingredients used in medicated soaps. Available from: https://www.cosmeticsdesign.com/Article/2016/10/20/Japan-bans-19-ingredientsused-in-soap
- Federal Register [Internet]. 2017 [cited 2024 Mar 1]. Safety and Effectiveness of Health Care Antiseptics; Topical Antimicrobial Drug Products for Over-the-Counter Human Use. Available from: https://www.federalregister.gov/documents/2017/12/20/2017-27317/safety-and-effectiveness-ofhealth-care-antiseptics-topical-antimicrobial-drug-products-for
- Yueh MF, Tukey RH. Triclosan: A Widespread Environmental Toxicant with Many Biological Effects. Annu Rev Pharmacol Toxicol. 2016;56:251–72.
- World Health Organization. Global guidelines for the prevention of surgical site infection [Internet]. 2nd ed. Geneva: World Health Organization; 2018 [cited 2024 Feb 12]. 184 p. Available from: https://iris.who.int/handle/10665/277399
- Berríos-Torres, Sandra I et al. “Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection, 2017.” JAMA surgery vol. 152,8 (2017): 784-791.
- Ademuyiwa AO, et al. Reducing surgical site infections in low-income and middle-income countries (FALCON): a pragmatic, multicentre, stratified, randomised controlled trial. The Lancet. 2021 Nov;398(10312):1687–99.
- World Health Organization. Global guidelines for the prevention of surgical site infection [Internet]. Geneva: World Health Organization; 2016 [cited 2024 Mar 1]. 184 p. Available from: https://iris.who.int/handle/10665/250680
- Quill® barbed suture IFU.pdf.